Page 32 - CITDBooklet
P. 32
Guide on the application of REACH Regulation
04REACH DATA
SHARING
4.1 Data sharing in SIEFs
■ Substance Information Exchange Forum (SIEF) – share relevant and available data
amongst all Potential Registrants of the same phase-in-substance.
■ SIEF would comprise of Manufacturers, Importers, Downstream users and other stake
holders
■ SIEF will be formed when potential registrants of a substance in the pre-registration list,
actually agree that they effectively manufacture, intend to manufacture or import a
substance that is sufficiently similar to allow a valid joint submission of data (Substance
identification process)
■ Aims of SIEF:
– Facilitate data sharing for purpose of registration thereby avoiding duplication
of studies
– Agree on the classification and labelling of the substance concerned
– SIEF is not a legal entity or a consortium. It is a forum to share data
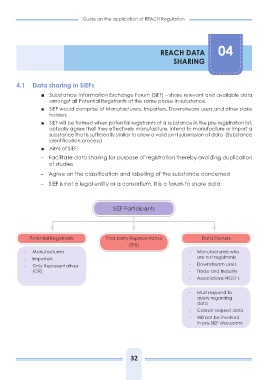
SIEF Participants
Potential Registrants Third party Representative Data Owners
(TPR)
- Manufacturers - Manufacturers who
- Importers are not registrants
- Only Representatives
- Downstream users
(OR) - Trade and Industry
- Associations-NGO’s
- Must respond to
query regarding
data
- Cannot request data
- Will not be involved
in pre-SIEF discussions
32