Page 34 - CITDBooklet
P. 34
Guide on the application of REACH Regulation
■ Manufacture and uses – current and foreseen,
■ Information on exposure-current and anticipated,
■ Information on Risk Management Measures – already implemented and proposed,
■ Data gathering should be irrespective of tonnage,
■ Registrant must register all relevant and available data – including data for higher
tonnage,
■ Participants may conduct literature search collectively.
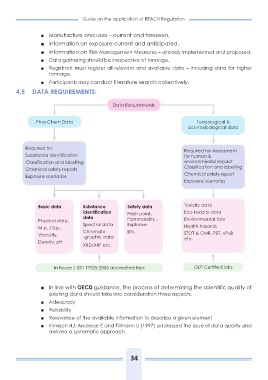
4.5 DATA REQUIREMENTS:
Data Requirements
Phys-Chem Data Toxicological &
Eco-toxicological data
Required for Required for Assessment
Substance identification for human &
Classification and labelling environmental impact
Chemical safety reports Classification and labelling
Exposure scenarios
Chemical safety report
Exposure scenarios
Basic data Substance Safety data Toxicity data
identification Eco-toxicity data
Physical state, data Flash point, Environmental fate
M.p. / B.p., Flammability , Health hazards
Viscosity, Spectral data Explosive STOT & CMR, PBT, vPvB
Density, pH etc.
Chromato Etc.
-graphic data
XRD/XRF etc.
In house / ISO 17025:2005 accredited labs GLP Certified labs
■ In line with OECD guidance, the process of determining the scientific quality of
existing data should take into consideration three aspects.
■ Adequacy
■ Reliability
■ Relevance of the available information to describe a given element
■ Klimisch HJ, Andreae E and Tillmann U (1997) addressed the issue of data quality and
derived a systematic approach.
34